T2 - Calibration¶
Models are simplifications of the real world, and quantities in the model (like the force of infection) represent the aggregation of many different factors. As a result, there can be uncertainty as to what value of the parameters most accurately reflects the real world - for instance, the population force of infection varies with the average number of contacts per person per day, but this quantity may not be well constrained. The first step in running a model is to improve estimates of the parameter values for a particular setting, using data from that setting. Typically, the model is started off at some point in the past (e.g. 2000), such that the initial compartment sizes correspond to the data in the simulation start year. The model is then run up to the current year, with the compartment sizes changing due to the model parameters. The model predictions can then be compared to the actual data for those same years. This allows model parameters to be adjusted to best match the existing data. These same parameters are then used for future projections.
To see calibration in effect, consider the following simple example:
[1]:
import atomica as at
P = at.Project(framework='assets/T2/t2_framework_1.xlsx',databook='assets/T2/t2_databook_1.xlsx', do_run=False)
First, we inspect the default calibration by running the model and plotting it along with the data. To plot the data, pass the project’s data to the plotting function (in this case, plot_series) - this will automatically add scatter points to the plot based on the data in the databook.
[2]:
result = P.run_sim()
d = at.PlotData(result,project=P)
at.plot_series(d, data=P.data);
Elapsed time for running "default": 0.0154s
Notice how the number of susceptible people and infected people exactly match the data points in the simulation start year - as noted above, this is because the model is initialized from the data values in that year. There are some conditions under which the model won’t exactly match the data in the initial year, such as if the initialization characteristics are overdetermined, but these situations are rare.
We can see, however, that the model does not predict enough susceptible people in 2020. There could be many reasons for this, and determining what parts of the model should be changed can often be something of an art. It typically reflects your understanding of the assumptions that were made in designing the framework, and also uncertainties and bias present in the input data. For example, the methodology used to gather data used for the calibration might provide hints as to which parameters to change first.
In this case, as there are insufficient people, it might be the case that the birth rate was too low. There are two ways to address this
You could go back to the databook and enter a larger value for the birth rate
You can add a ‘scale factor’ to the parameter set, which scales the parameter value up or down
Either approach can be used and would provide equivalent results. Why would we prefer one over the other?
Decision factor | Databook calibration | Scale factor calibration |
|---|---|---|
How do you want to adjust the parameter? | Manual adjustment | Automatic adjustment |
What kinds of parameters is this appropriate for? | Appropriate for model assumptions | Appropriate for original data |
Granularity of calibration? | Adjustments can vary by year or even timestep | Single scaling factor for all timesteps |
Pros? |
|
|
Cons? | Can cause confusion in the databook around what is data and what is not data | Can lack transparency about how parameters are being adjusted without careful review |
An example of a suitable parameter for databook calibration is relative seasonality of mosquito population size - it is unlikely that any existing data would be able to directly provide a value for this parameter, but it might be possible to calculate proxy values from other data such as rainfall patterns in different years, adjust annual values manually to match historical epidemic patterns, and then assume a median value for future years. Having this assumption in the databook allows for
those calculations to be used as a starting point in the databook before manually editing, and allows for comparability with other projects that use the same parameter.
Suggestion: When designing a databook, it is recommended that all parameters intended for explicit databook calibration are placed on a single ‘Calibration’ sheet to provide clarity about what is data and what are calibrated assumptions.
An example of a suitable parameter for scale factor calibration is treatment initiation used to determine model transitions from diagnosed to treated - a country has reported data for the number of people initiating treatment, and it is important to accurately maintain the official numbers in the databook. Nevertheless, there may be systematic under-reporting by an unknown degree and we want to account for those additional treatments in the model to ensure outcomes are representative, so it
is appropriate to adjust the scale factor.
An example of a parameter that could be adjusted in either way depending on the circumstances or just personal preference would be force of infection - this is a clear calibration parameter that is not based on data, and could be adjusted in a databook if it’s necessary to reflect changing circumstances external to the model over time, calibrated automatically with a scale factor via the calibrate function below in order to achieve the best fit to data, or even a mixture of the two
methods.
Calibration scale factors¶
The values in the databook are intended to serve as a record of the original input data used to inform the model. Therefore, if a parameter needs to be scaled up or down to calibrate the model, it is typically preferable to do this by setting a ‘scale factor’ (also referred to as a ‘y-factor’) in the ParameterSet, rather than changing the data.
The web interfaces (such as the Cascade Analysis Tool) perform calibration using scale factors. The scale factors shown on the website correspond to the values being set here.
To set a scale factor, create a ParameterSet either by copying an existing one, or by creating a new one. Then, access the pars attribute to look up the parameter you wish to change, and set the y_factor for the population you want to change:
[3]:
p2 = P.parsets[0].copy() # Copy an existing ParameterSet
p2.pars['b_rate'].y_factor['adults'] = 2
The example above doubled the birth rate. Now we can run the model again, and see how the results have changed. Notice how the PlotData command is being called with both the original results object, and the new results object, allowing both model runs to be shown on the same plot.
[4]:
r2 = P.run_sim(parset=p2,result_name = 'More births')
d = at.PlotData([result,r2], outputs='sus',project=P)
at.plot_series(d,axis='results',data=P.data);
Elapsed time for running "default": 0.0143s
We can see that we have considerably overshot the data, indicating that doubling the birth rate was much too big a change. This would typically be the first step in an iterative process, where you adjust the scale factor, inspect the data, and then make further adjustments.
Calibrating compartment sizes¶
The ParameterSet contains parameters for quantities entered on the ‘Compartments’ and ‘Characteristics’ sheet of the databook. These are used to calculate the initial compartment sizes used at the start of the simulation. Y-factors can be set for these quantities, the same as for any other parameter. This will not change the data points - just the calculated initial compartment size. For example, we can initialize the model with half as many susceptible people by setting a y-factor of 0.5 on
the sus compartment size:
[5]:
p2 = at.ParameterSet(P.framework, P.data) # Make a new ParameterSet
p2.pars['sus'].y_factor['adults'] = 0.5
r2 = P.run_sim(parset=p2,result_name = 'Decreased initial sus')
d = at.PlotData([result,r2], outputs='sus',project=P)
at.plot_series(d,axis='results',data=P.data);
Elapsed time for running "default": 0.0126s
Unlike setting y-factors for parameters, y-factors for compartment sizes may result in invalid initializations. This can occur if the scaled compartment sizes or characteristics are inconsistent with each other. For example, if the calibrated number of initial susceptible people results in needing more people than the total population size, an error will occur:
[6]:
p2 = at.ParameterSet(P.framework, P.data) # Make a new ParameterSet
p2.pars['sus'].y_factor['adults'] = 1.1
try:
r2 = P.run_sim(parset=p2,result_name = 'Decreased initial sus')
except Exception as e:
print(e)
Negative initial popsizes:
Compartment adults rec - Calculated -100.000000
Characteristic 'alive': Target value = [1500.]
Compartment sus: Computed value = 1100.000000
Compartment inf: Computed value = 500.000000
Compartment rec: Computed value = -100.000000
In this case, such an error could be resolved by also increasing the y-factor for the ‘alive’ characteristic, thereby increasing the total population size:
[7]:
p2 = at.ParameterSet(P.framework, P.data) # Make a new ParameterSet
p2.pars['sus'].y_factor['adults'] = 1.1
p2.pars['alive'].y_factor['adults'] = 1.2
r2 = P.run_sim(parset=p2,result_name = 'Increased initial sus')
d = at.PlotData([result,r2], outputs=['sus','alive'],project=P)
at.plot_series(d,axis='results',data=P.data);
Elapsed time for running "default": 0.0118s
See https://atomica.tools/docs/master/general/Compartment-Initialization.html for more information on how the initialization of compartment sizes is carried out by Atomica.
Calibrating transfers¶
It is also possible to set y-factors to calibrate transfers, that move people from one population to another. A transfer moves people from one population to another e.g., aging from 5-14 to 15-64. Therefore, a y-factor for a transfer must be identified by two populations rather than one. Further, the transfer parameter does not appear in the framework, because they are population-specific. Instead, the transfer is defined in the databook, and then a corresponding parameter is automatically
created in the ParameterSet. For example, we can load the TB demo in Atomica and inspect the transfers:
[8]:
P2 = at.demo('tb')
Elapsed time for running "default": 0.625s
In the databook, there are two transfers, relating to aging and incarceration:
[9]:
P2.data.transfers
[9]:
[<TDC Transfer "age">, <TDC Transfer "inc">]
These both appear in the ParameterSet under the ‘transfers’ attribute:
[10]:
P2.parsets[0].transfers.keys()
[10]:
['age', 'inc']
To set a scale factor, it is necessary to define both the source and destination population. The transfer is indexed first by the source population, and then by the ‘to’ population. The top level key in the transfer corresponds to the source population:
[11]:
P2.parsets[0].transfers['age'].keys() # Source populations
[11]:
['0-4', '5-14', '15-64']
If we index the transfers using this code name, we can access a Parameter that contains y_factor entries for all of the destination populations.
[12]:
P2.parsets[0].transfers['age']['5-14']
[12]:
<atomica.parameters.Parameter at 0x7fceb89b9c70>
[<class 'atomica.parameters.Parameter'>, <class 'atomica.utils.NamedItem'>]
————————————————————————————————————————————————————————————————————————
Methods:
copy() interpolate() smooth()
has_values() sample()
————————————————————————————————————————————————————————————————————————
Properties:
pops
————————————————————————————————————————————————————————————————————————
_interpolation_method: 'linear'
created: datetime.datetime(2025, 6, 18, 5, 10, 43,
854743, tzinfo=tz [...]
meta_y_factor: 1.0
modified: datetime.datetime(2025, 6, 18, 5, 10, 43,
854751, tzinfo=tz [...]
name: 'age_from_5-14'
skip_function: #0: '15-64': None
ts: {'15-64': <atomica.utils.TimeSeries at
0x7fceb471f6a0>
[<cl [...]
y_factor: #0: '15-64': 1.0
————————————————————————————————————————————————————————————————————————
Notice how this parameter only contains y-factors for the destination populations associated with the ‘age’ transfer out of the ‘5-14’ population
[13]:
P2.parsets[0].transfers['age']['5-14'].y_factor
#0: '15-64': 1.0
[13]:
This corresponds to the fact that the aging transfer can only move people in 5-14 population to the 15-64 population. To set a y-factor for this transfer, assign a value to this entry in the y_factor attribute:
[14]:
P2.parsets[0].transfers['age']['5-14'].y_factor['15-64'] = 1.5
The example above will increase the transfer rate for aging from 5-14 to 15-64.
Automatic calibration¶
Automated calibration is also available via the project’s calibrate method. This will automatically adjust parameter values to match the data. To use this function, you need to specify which parameters to set scale factors for, and which variables in the databook to compute calibration quality from. The framework can provide defaults for which parameters to automatically calibrate, or you can pass a list of those parameters in directly. In this example, we will pass in b_rate because we
want to adjust the birth rate, and we will use sus as a measurable since we want to match the number of susceptible people. The configuration therefore corresponds to the example shown above.
[15]:
with at.Quiet():
p3 = P.calibrate(max_time=10, parset='default', adjustables=['b_rate'], measurables=['sus']);
The result of automated calibration is another ParameterSet. We can inspect the scale factor that the algorithm found:
[16]:
p3.pars['b_rate'].y_factor
#0: 'adults': 1.318694543838501
[16]:
and we can run the model to compare the automated calibration to the original default calibration:
[17]:
r3 = P.run_sim(parset=p3,result_name = 'Auto calibration')
d = at.PlotData([result,r3], outputs='sus',project=P)
at.plot_series(d,axis='results',data=P.data);
Elapsed time for running "default": 0.0114s
Automated calibration of transfers¶
As described above, transfers are identified by two populations rather than one. The ‘adjustable’ for a transfer parameter is therefore identified by concatenating the name of the transfer and the name of the source population, separated by '_from_'. For example, we could specify an adjustable of 'age_from_5-14' to calibrate the aging transfer out of the 5-14 population.
Calibration tips¶
While calibrations can vary significantly from model-to-model, it’s generally a good idea to try and match coarse-grained quantities first, followed by fine-grained quantities. For example, for TB you might calibrate it in the following order:
Match population size (adjusting birth rate and death rate)
Match disease prevalance (adjusting force of infection)
Match drug-resistant/drug-susceptible split (adjusting proportion of infections that are drug-resistant)
For complex models when considering how to proceed with a calibration, it can help to start with mapping the expected relationships between key input parameters that will be used for calibration and key output parameters for which data exists and that should be matched by the calibration, in terms of how changes might flow throughout the model.
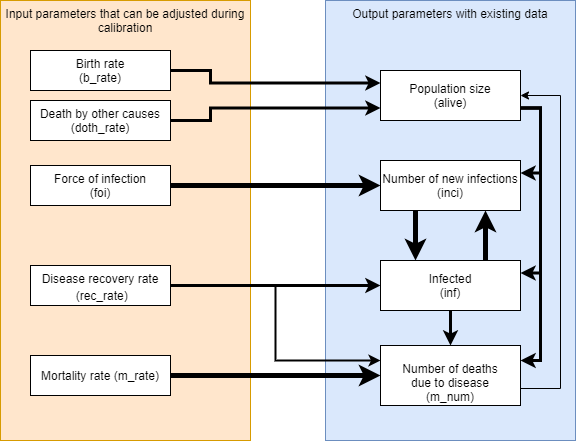
From the diagram above, it can be seen that as is typically the case, population size (alive) has a large impact on everything else, but the number of disease-related deaths have only a minor impact on population size in return, so population size needs to be matched first. Incidence (inci) and prevalence (inf) have a strong cyclical relationship should be considered together, but force of infection and recovery rate have direct links to modifying each of those individually through
changing the rate at which people are infected and changing the rate at which people recover. Disease-related deaths (m_num) can be considered last if it is not already closely matched from calibrating to prevalence. Because this may adjust the population size, an iterative cycle of calibration may be appropriate to get the best overall fit.
A calibration might then proceed in the following order with three repeats:
[18]:
cal_par = P.parsets['default'].copy()
with at.Quiet():
for _ in range(3):
cal_par = P.calibrate(max_time=10, parset=cal_par, adjustables=['b_rate', 'doth_rate'], measurables=['alive'])
cal_par = P.calibrate(max_time=10, parset=cal_par, adjustables=['foi', 'rec_rate'], measurables=['inci', 'inf'])
cal_par = P.calibrate(max_time=10, parset=cal_par, adjustables=['m_rate'], measurables=['m_num'])
[19]:
r4 = P.run_sim(parset=cal_par,result_name = 'Auto calibration repeated')
for output in ['alive', 'inci', 'inf', 'm_num']:
d = at.PlotData([result,r4], outputs=output,project=P)
at.plot_series(d,axis='results',data=P.data);
Elapsed time for running "default": 0.0106s
There are a few major dangers in calibration, including but not limited to:
1. Solution-rich spaces
Often multiple input parameters can be adjusted and they might superficially produce the same historical trend lines. For example if modelled prevalence is far lower than data points from epidemiological surveys, a better calibration might be achieved in a number of different ways: increased force of infection (calibration parameter), increased behavioural risk factors (e.g. number of shared injections for HIV), increased duration of infection, reduced treatment success rate, or any number of other subtle data inputs. Where possible calibrate to include data from multiple other outputs, and even if this is not possible review other outputs as a sanity check. In addition, consult country experts to determine which solution better explains the trend where it is not clear from the data.
2. Overfitting
Don’t try to exactly match every historical data point such as by adjusting ‘force of infection’ precisely for every historical year. Often there is year to year variation in diagnostic capacity, reporting, or natural fluctation based on behaviour or external circumstances. It is more important to accurately capture the trend and the reasons for that trend in the model than to match data from every year exactly, and this will lead to more reasonable future projections as well as a more useful model for prioritizing programs.
3. Inaccurate data
In conjunction with overfitting - not all data is of equal quality or reliability, or it may have been gathered with a methodology that does not exactly fit the way it is used in the model (this is often a good argument to adjust the model if it applies to more than a single project). Be aware of the quality of data and prioritize calibration to data that is known to be more accurate in a given project. Sometimes it is better to just ignore data for a specific output parameter if the methodology used to gather that data was unreliable or cannot be adequately represented by the model.
4. Forced outcomes
Especially with automatic calibration, it is possible to match outputs with some extreme values for y_factor. Some examples of how this can occur:
The calibration of
previn a Males 50+ population using thefoiparameter results in ay_factorof 0.00001 - if optimizations are run with this force of infection value, the Males 50+ population will be almost immune to infection for any reason, and any programs that target Males 50+ will be defunded. In reality, something else is wrong in the model, perhaps this high prevalence is because of Males 40-49 aging into the 50+ population and that population should be reviewed, or even more likely there is a risk-based transition from a key population such as people who inject drugs which is too high.The calibration to match
incidencein children aged 0-14 using thefoiparameter results in ay_factorof 1000. The real reason for this may be that the model itself has failed to include the critical pathway for child incidence through mother-to-child transmission.
In order to avoid these kinds of modelling outcomes it is critical to (a) review calibration values/y_factors to ensure they are within expected bounds, and (b) if there are extreme values conduct further investigation, as there will be something else that should be changed to improve how the model represents reality, and this will result in better recommendations using the model.
[20]:
for par in ['b_rate', 'doth_rate', 'foi', 'rec_rate', 'm_rate']:
print (f'{par}: y_factor = {cal_par.pars[par].y_factor["adults"]}')
b_rate: y_factor = 1.0646636962890628
doth_rate: y_factor = 0.2414290566194772
foi: y_factor = 1.3290953683648261
rec_rate: y_factor = 0.11732962890145406
m_rate: y_factor = 0.12464730441570263
Some of these values are low at around 0.15, and this can be attributed to the high value for prevalence (inf) in the databook requiring a low value for each of doth_rate, rec_rate, and m_rate. Individually these are not outside of a ‘reasonable’ range for calibration y_factors but as they are all needed to fit a single data point this might suggest a second look at the reliability of original data values for prevalence in this project.
5. Missing model features Calibration at its best accounts for factors outside of a model in order to focus on what is in the model. Sometimes those factors have too big an impact on the model to be ignored, and the right solution is to add depth to the model as the only other solution is to force outcomes with extreme y_factors or unrealistic values for other parameters.
In the example above, it is impossible to match both the 2010 and 2020 data points for number of deaths (m_num) as they are not consistent with increasing prevalence. Perhaps it is necessary to calibrate a changing m_rate over time in the databook, or even add diagnosis and treatment to the model?
6. ‘Burn-in’ period Often with more complex models, there may be too many parameters to initialize each and every one of them accurately in the first time step of the model via databook entry, such as tracking compartments for many different stages of a disease or stages of treatment. In these cases, it may be best to initialize just using key databook values such as ‘number on treatment’, ‘number infected’ and make assumptions for compartments within these categories. This will typically result in strange model behaviour for a number of time steps during a ‘burn-in’ period before the proportions at different stages of treatment settle down to an equilibrium, so it may be necessary to exclude these burn-in years from exported results in general, and not try to match these in calibration. Ideally, the model should either initialize a model earlier (e.g. several years in models with long time steps) and/or calibrate to the years for which data exists, and exclude those burn-in years from exported results in general.